What makes kveik a super-yeast

Microbiology lab at University of Guelph. |
When Richard Preiss first started studying kveik his former thesis advisor at the University of Guelph, George van der Merwe, told him that it was a "super-yeast" because of its speed and stress resistance. The lab that van der Merwe heads at the university has been investigating how yeast responds to stress for many years, and that made them extra excited about kveik. Since kveik was so robust, maybe it knew some special tricks for handling stress?
Several years in the making, van der Merwe's group now has a preprint out that unravels some of the secrets behind kveik's ability to handle many types of stress. Richard describes the preprint as "a bit of a monster," and that it certainly is, but I'll try to explain and unpack what is going on here so that it becomes understandable. There's so much going on that I'm going to focus on the main result here, and leave the rest for a second post.
When I say kveik is unusually robust, what do I actually mean?
- It can handle higher temperatures than most yeasts.
- It has higher alcohol tolerance (13-16%) than most yeast (8-10%).
- It can be dried (that's unusual). Richard once remarked that kveik dried by farmhouse brewers at home was in many cases healthier after rehydration than dried yeast from multi-billion-dollar companies.
- It can be frozen (also unusual).
- It can probably handle higher sugar concentrations than most yeasts (not documented, but I'm pretty sure it's the case).
- It can be stored for longer than most yeasts.
In addition it ferments faster than other yeasts, so when George van der Merwe called it a "super-yeast" he was not joking.
So what's the secret?
It's not hugely complicated, but for the answer to make sense we need to explain a few things first.
The lipid bilayer
All living cells have a cell membrane, which is crucial because it separates the inside of the cell from the outside. This separation enables the cell to have its own special biochemistry on the inside, and that biochemistry is basically what life is. It also allows the cell to control which substances come into the cell and which ones go out of it.
|
Cell membrane diagram. (By Ladyofhats, from Wikimedia Commons.) |
Curiously, the membrane is basically built the same way in all organisms: from a lipid bilayer. Look at the figure above. See the red dots with yellow tails? Those are lipids (fatty acids) which have a special property: their heads want to attach to water, and their tails want to avoid water. The amazing consequence is that in water these particular lipids line up in a double layer (bilayer) with the tails facing each other (away from the surrounding water).
To make a simple cell membrane it's basically enough to make a lot of these lipids and they'll form into a protective membrane all by themselves. That obviously has huge implications for how life came about originally.
As you can see in the diagram a full membrane is a bit more complicated than just a simple lipid bilayer, since it has various components sitting in it. Particularly important for us are the ports (far left, "protein channel") that can pull substances into the cell, or push them out.
Note that the cell wall is not the same as the membrane. The cell wall is a tough support layer on the outside of the membrane making it stronger, and not all organisms have it. Animals, for example, such as you and me, don't.
Threats
Weirdly, a lot of the things that endanger yeast cells turn out to revolve around the same thing: the stability of the cell membrane.
The cell membrane is self-organizing in water, but what happens if you remove much of the water? The answer is that the lipid bilayer becomes unstable because the self-organization doesn't quite work any more, and it can start leaking, or even fall apart.
Obviously, this will happen if you dry the yeast, but, intriguingly, adding too much sugar or too much alcohol has much the same effect. Both because this means there is necessarily less water percentage-wise, but also because sugar and alcohol can disrupt the membrane. The sugar causes osmotic pressure against the membrane, and too much of that is hard to handle.
It's the same thing with temperature: if the temperature is too low or too high the self-organization of the lipids stops working as well and the cell wall can start leaking, which causes all sorts of problems.
In addition, alcohol can cause problems internally in the cell, by making other substances more toxic. High temperatures will, once they get high enough, start destroying crucial proteins inside the cell. This can include both structural components in the cell and enzymes making things happen, so that really causes things to fall apart.
But how does kveik counter these problems?
The super-weapon
The answer is trehalose. Kveik makes lots of trehalose.
If that didn't make any sense to you, be glad we just went through all this initial biochemistry stuff, because that means we're in a position to understand what's going on.
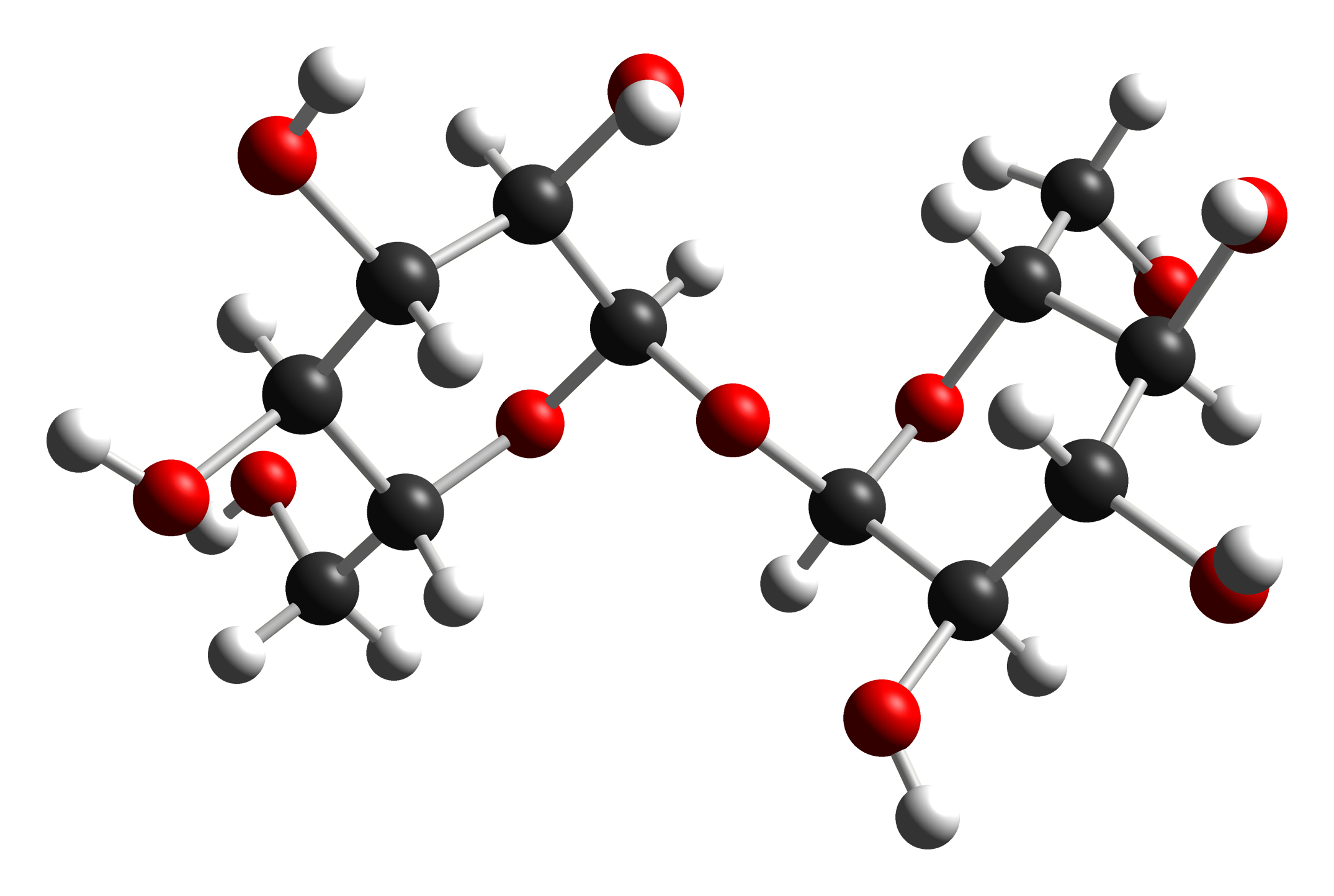
The molecular structure of trehalose. (Ben Mills, from Wikimedia Commons.) |
Trehalose is a pretty simple sugar: it's formed from two glucose molecules bonded together. That's what you see in the diagram: two glucose molecules tied together with one oxygen atom (red dot). Glucose is one of the simplest sugars and there's a lot of it in honey. Table sugar, on the other hand, is made of sucrose, which is formed by bonding together one glucose and one fructose molecule.
But how can a sugar molecule ward off all these threats?
It's actually incredibly simple: trehalose can keep the cell wall together by replacing the water. Trehalose will stick to the cell wall and make sure the lipids stay in place. For it to be really effective you need trehalose on both sides, but this can be solved by having one of those transporters that can pump trehalose out of the cell.
Trehalose can also stabilize the precious proteins inside the cell, probably using a similar trick. So it helps not just with the membrane, but also internally.
It's actually not just yeasts which use trehalose: many species of bacteria, insects, plants, invertebrate animals, and other fungi protect themselves this way.
But how did the researchers show that kveik actually uses trehalose?
The actual results
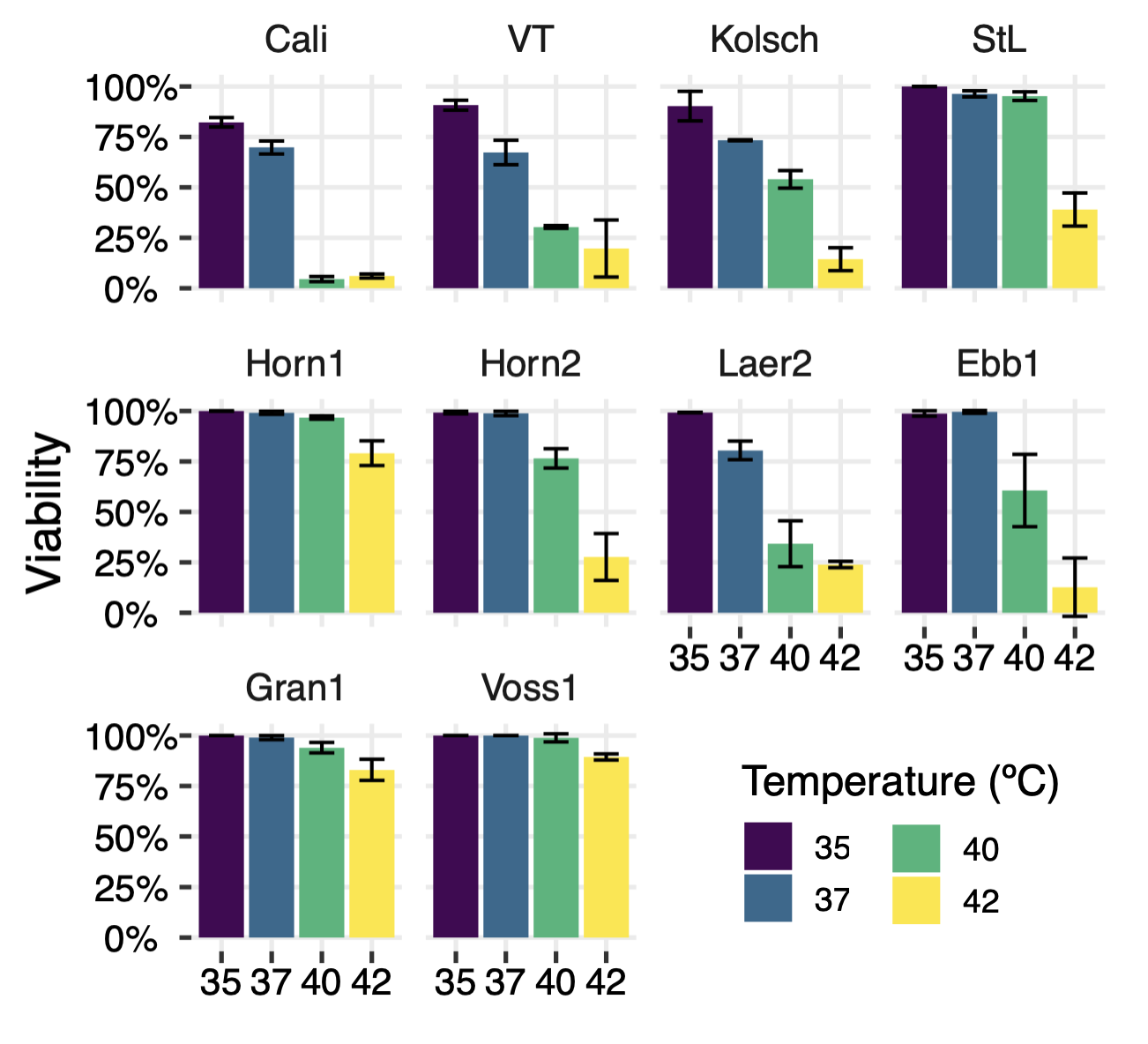
What percentage of cells is still alive after growing different yeast strains at different temperatures. Figure 5 from the paper. |
The researchers grew different yeast strains and different temperatures, then measured what percent of cells were still alive. The diagram above has the results. The lower two rows are kveik strains, and you can see they all handled 35-37C fine. Some struggled with 40C and/or 42C, and the Voss and Granvin strains were fine at all temperatures.
The top row is the control strains. The first three, WLP 001 California Ale Yeast, Vermont Ale Yeast, WLP 029 Kölsch Ale Yeast, all struggle at all these temperatures. The last one, St. Lucifer, is more like a kveik, although it too struggles at 42C.
Thus far, this is stuff pretty much everyone knows: kveik can handle higher temperatures than other yeasts. There are two twists, though: some Belgian yeasts are about as thermotolerant. And some kveik strains are not great at the highest temperatures. Note that this last point doesn't necessarily mean the original culture would have problems at these temperatures, because it might contain other strains that would handle the temperature just fine. (Do they? We don't know.)
But what about the trehalose? Well, let's look at one more figure.
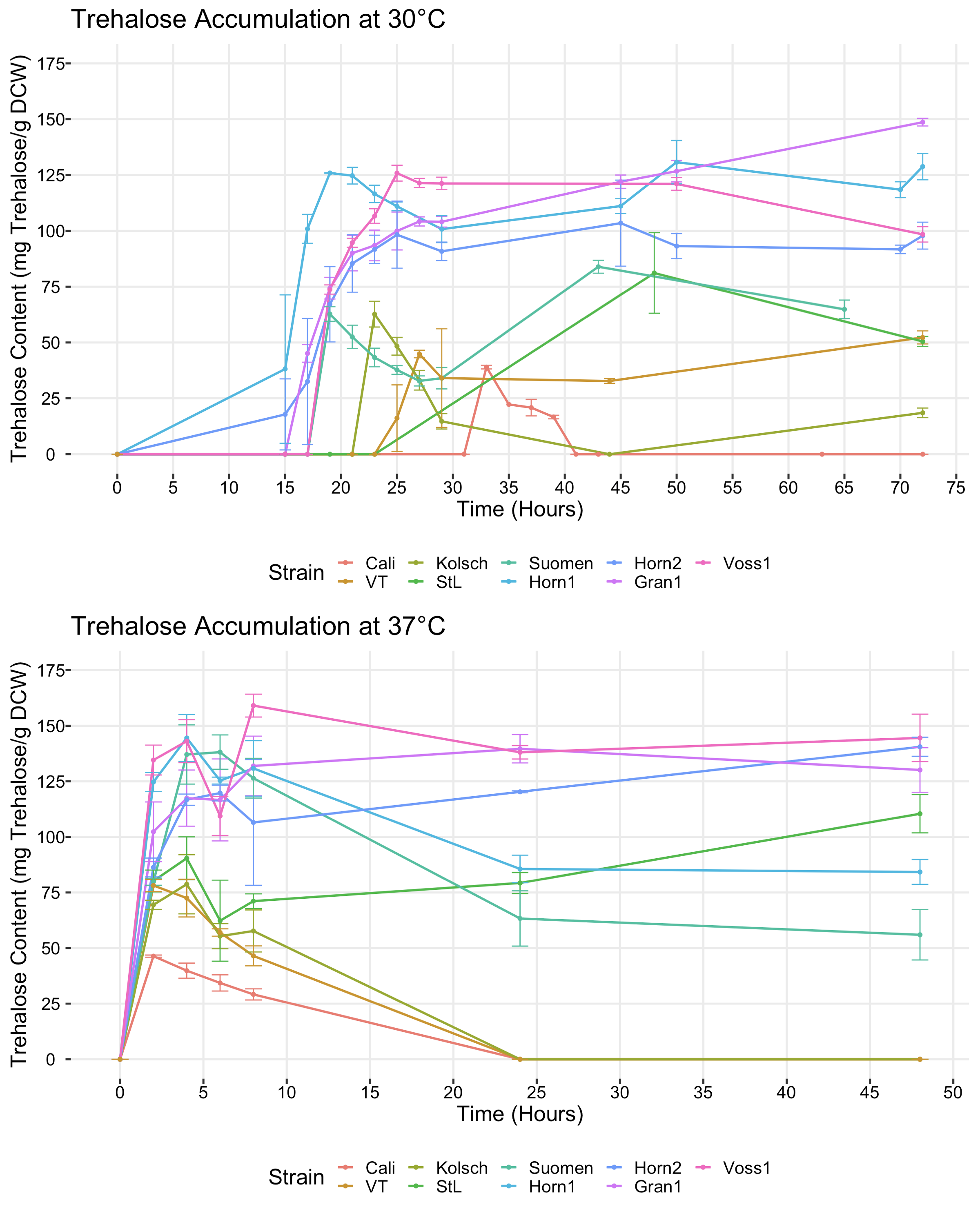
Trehalose content over time at two different temperatures. Figure 6 from the paper. |
The key point is that at 30C (top diagram) none of the strains make trehalose until about 15 hours after growth begins. But if we increase the temperature to 37C (bottom diagram) suddenly all the strains start making trehalose right away. So clearly the kveiks (and other strains) use trehalose to protect themselves against heat. And the kveiks make much more of it than the other strains.
Another interesting thing is that the non-kveiks consume much of their trehalose again, while the kveiks leave it hanging around. Even when fermented at 30C the kveiks eventually make trehalose, even though there is no stress that forces them to. The reason is that the kveiks come from Norwegian farmhouse brewing, where the yeast was dried after harvesting, so for them keeping the trehalose was obviously important. The control strains would expect to be kept wet for a short while, then repitched into new wort, so for them trehalose would be much less important.
But if making trehalose turns you into a super-yeast, why don't all yeast strains do it? There's an answer to that, too.
No free lunch
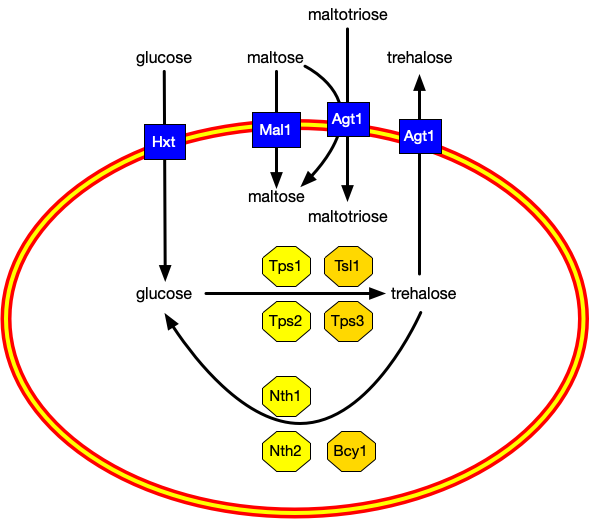
How trehalose is produced, exported, and broken down. |
Remember how trehalose had to be on both sides of the membrane to protect it fully? To get these big molecules through the membrane the yeast needs a transporter, and the AGT1 gene makes a transporter that sends trehalose out of the cell, as you see in the diagram above.
The seven octagons are genes. Let's start with the upper four: the yellow ones turn glucose into trehalose inside the cell, and the orange ones are regulatory genes that control when trehalose is produced. The kveik strains have changes in all four of these genes, but particularly in the regulatory ones. That's very likely the reason they make more trehalose.
The genetics also seem to explain why the kveiks retain more trehalose than ordinary yeast: they have more copies of the Bcy1 gene, which regulates how trehalose is broken down back to glucose. The details are a bit involved, but essentially that seems to be the genetic reason why they retain the trehalose.
But I said there was a catch. To explain it we have to do a little detour.
As you know, there's many kinds of sugar in wort. Fructose, glucose, and sucrose make up about 30%. A whopping 45% is maltose, which, like trehalose, is made from two glucose molecules, but with a different bond between them. There's also about 20% maltotriose, which is three glucose molecules, with the exact same bond between them as in maltose.
There are ports pulling all of these various sugars into the cell, as you would expect. What you probably wouldn't expect is that the port for importing maltotriose is the same one that's exporting trehalose: Agt1. See the diagram.
It seems that in order to efficiently export trehalose to get all this super robustness, kveik ended up changing Agt1 so that it works better for pushing trehalose out and worse for pulling maltotriose in. The researchers demonstrated that by testing how much of the maltotriose gets fermented, as shown in this figure:
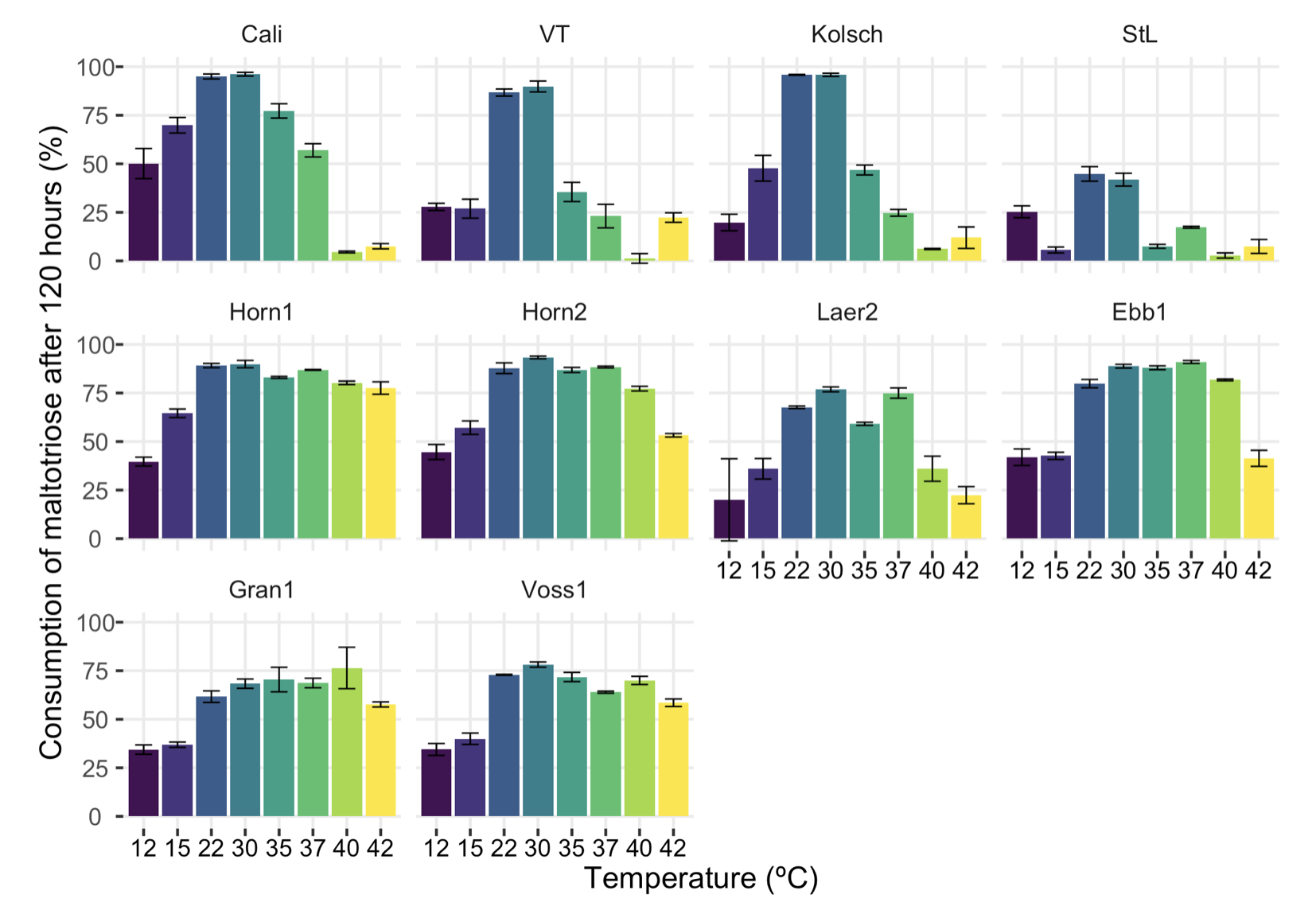
Percent of maltotriose consumed by different strains at different temperatures. Figure S2 from the paper. |
Now, I guess anyone looking at this figure for the first time will conclude that it shows the opposite: that kveik ferments more maltotriose than the control strains. But what you're actually seeing is that kveik is more thermotolerant than the controls, which you already knew. That is, the kveiks have higher bars at a broader range of temperatures.
Look at the highest bar for each strain, where the yeast is fermenting as much maltotriose as it possibly can. The three "normal" controls can ferment 80-95% of the maltotriose when conditions are right, while half the kveiks can't get above 75%, and even at the optimum they don't get higher than 85%.
But the farmhouse brewers usually ferment down to fairly reasonable final gravities, so how does that work?
The tradition works
The percentage of wort made up by the various types of sugar is not the same in every wort: it depends on how you mashed. If you do a normal craft brewing mash of 1 hour at 67C you'll get pretty close to the percentages I gave above. In that case you can indeed end up with a fairly high FG, because the kveik strain you use may be a poor fermenter of maltotriose.
So if your kveik fermentations end up with an FG that's higher than you wanted, this could be the reason. You may want to look at your mashing to reduce the maltotriose in your wort.
But look back at that first meeting with Sigmund Gjernes: he mashed for six hours. That gave the enzymes in the mash plenty of time to cut the maltotriose (3x glucose) down to maltose (2x glucose). The normal in Voss is four hours, which has pretty much the same effect.
And the raw ale brewers further north never boil, so their malt enzymes keep working in the fermentor. Yes, they work much slower at 30C, but they have 2-3 days to finish cutting down the maltotriose.
Whether kveik has adapted to the tradition or the tradition has adapted to the kveik is hard to say. It's notable that the brewers I know in Sogn who don't use kveik have shorter mashes.
What is clear is that although traditional brewers may not know why they do what they do, they're surprisingly often right to do what they do.
Acknowledgements
I want to make it very clear that this is not my work. I'm just repeating what's in the paper, and I had a bit of help from Richard Preiss with that.
The people who did the actual work are (in the order given in the paper):
- Barret Foster (University of Guelph),
- Caroline Tyrawa (University of Guelph),
- Emine Ozsahin (University of Guelph),
- Mark Lubberts (University of Guelph),
- Kristoffer Krogerus (VTT in Finland),
- Richard Preiss (Escarpment Labs),
- George van der Merwe (University of Guelph)
Similar posts
There is a Whole Family of European Farmhouse Yeasts
Six years ago, the paper that told us where kveik came from was published
Read | 2024-04-07 14:47
Where kveik comes from
I've written before about the kveik research paper by Preiss, Tyrawa, and van der Merwe
Read | 2018-09-12 16:27
A family tree for kveik
In 2016 I was contacted by Canadian researcher Richard Preiss
Read | 2017-10-06 10:02
Comments
Vernon Tafte - 2021-08-09 14:49:50
Hello I have been experimenting with kveik Voss i’m still having trouble getting the yeast to go all the way dry starting gravity is 1.096 . Can you tell me how much nutrient this strain needs to go all the way thank you Vernon
James - 2021-08-09 17:21:13
It seems as though kveik’s reliance on trehalose outside the cell would make it vulnerable to other microbes that consume trehalose. In other words, it’s all well and good to pump trehalose into the beer to reduce stress, but if another yeast or a bacteria is gobbling up the trehalose then it presumably won’t do much good (obviously it’s a different story for the trehalose inside the cell).
I wonder if kveik evolved to ferment so quickly partly so that it could finish the job and get dried before competing microbes have a chance to deplete the trehalose.
Lars Marius - 2021-08-09 19:02:18
@Vernon: What's "all the way dry"? What FG are you looking for? With 1.096 OG you don't need to worry about nutrient. Maybe you're underpitching too much, or the yeast is too old? Maybe there isn't enough oxygen? Or maybe, like this blog post talks about, you're not mashing long enough? Or your mash temperature is off?
There's a thousand variables, unfortunately, so just knowing your OG isn't enough.
Lars Marius - 2021-08-09 19:03:55
@James: The trehalose isn't aimlessly pumped into the beer, though. If it were, it would be too diluted to be of any use. Instead, it sticks to the outside of the cell membrane. So I don't think having it consumed by other microorganisms is a concern.
I do think kveik (and other farmhouse yeast) evolved to be quick in order to be harvested before time was up. Although I don't think that has anything to do with the trehalose. I'll write a bit more about this in the next post.
Luke - 2021-08-09 23:41:59
Just wanted to say this a really great example of science communication. Well done!
James - 2021-08-11 01:45:23
Thanks Lars, I didn't realize the trehalose sticks to the cell. It's great to see progress being made in understanding how kveik works.
Mads Leonard Holvik - 2021-09-29 13:32:14
Great article! I was wondering if the kveik's use of trehalose is more at critical moments of high or low temperature or other stressors? Or is the kveik using trehalose all the time during fermentation equally?
Lars Marius - 2021-10-06 09:13:20
@Mads: Look at the graphs showing trehalose over time (Figure 6). You can see that the kveik is making trehalose in response to the high temperature (37C).
But it seems like kveik eventually makes trehalose no matter what, and the reason is probably that it expects to be dried after fermentation, and that it needs to be ready for that.
Stella - 2022-03-15 10:19:29
Hi Lars
Thank you so much for your in-depth articles about Kveik, based on scientific research. Very interesting and easy to understand.
I've been introducing three Kveik strains to the craft brewery where I'm writing my thesis (about Kveik btw), and I've been experiencing some very strong sulfury off-flavors the last two times, as I increased the fermentation temperature up to 37C. Nothing else in the brewing process has changed. I didn't add any nutrients, neither before nor now, and before I didn't experience any unwanted taste/aroma. It might be far out, but can these off-flavors be related to anything you mentioned in this post?
Lars Marius - 2022-03-15 12:16:38
@Stella: There have been anecdotal reports of sulphury flavours from kveik, but never with enough detail that it's been possible to figure out what's going on.
I don't know what you were fermenting, so it's hard to say whether nutrients were the problem or not. They could be.
To me it seems the most likely cause of problems would be contamination by yeasts other than kveik. I can't imagine any real kveik giving off-flavours simply because it ferments at 37C.
But without more data it's impossible to say.
Björn Petersson - 2022-03-17 16:56:23
The characteristics of kveik has developed by the use for centuries. But did they use the same yeast for baking and have that any importance for the special characteristics kveik got? When baking the temperature of the liquid is often between 30-40 C and you want a fast development of carbondioxide. Sourdough was also used for baking but "jästkrans, jäststock, jästkubb" etc. was used all over Scandinavia for drying and storing the yeast both for baking and brewing. The artisanal brewers on Gotland nowadays use bakers yeast and also some whisky distillers do. Scientific comparisons have been made between kveiks and other types of brewing yeasts but I have not seen that with bakers yeast That would be interresting.
Lars Marius - 2022-03-17 17:14:55
@Björn: Those who baked leavened bread nearly always used the yeast also in baking, yes. But yeast that goes into a bread then is baked and dies. So it's the beer yeast that lives on.
Body temperature was the normal temperature both for farmhouse ale fermentation and for raising bread.
Yes, these devices were used to store yeast, but generally beer yeast and not sourdough.
The Gallone 2016 and also later yeast phylogenetics papers do have some work on bread yeast, but not as much as one would wish. You may want to read this, if you haven't already https://www.garshol.priv.no/blog/411.html
Sage - 2022-08-10 21:30:57
Is there any anecdotal evidence that the higher levels of trehelose have an impact on flavor perception?
Is it eventually consumed in the fermentation process or do the cells hold on to it until they die off?
I did a quick google search and only saw some information related to salt and trehelose whereby changing the type of salt and increasing trehelose impacted (reduced) bitterness perception.
Lars Marius - 2022-08-10 22:06:23
@Sage: There is no indication that anyone can taste the trehalose, no. The measured amounts were taken from inside the cells and amount to 10% of the total protein weight of the cells.
If the amount of trehalose on the outside of the cells is the same then I assume that would be far too little for anyone to taste.
As you can see from the graphs, ordinary yeast consumes the trehalose again, while the kveiks do not. I presume the kveiks are keeping the trehalose because they expect to be dried and they want to survive that. So they may actually keep it around indefinitely.
Trevor Stimson - 2025-01-17 19:38:26
Hey Lars Loving your research and really getting into kveik style fermentation. I am a distiller in America and have made some awesome products with kveik yeast. Your book gave me an idea to apply selective pressures on wild caught and commercial yeast strains to steer evolution towards kveik like characteristics. The pressures I'm considering include high pitching and fermentation temps, drying, and freezing. Are there any more pressures I could add?